 
There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.\). Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. You can see that The molar mass of Magnesium is 24.305 g/mol. The relative atomic mass is the same as the molar mass (except molar mass is in g/mol). For example, if we are trying calculate for ammonia (NH 3 ), then we. The atomic mass is equal to the atomic number which is listed below the element symbol. 
So let’s look at the molar mass of Magnesium and Chlorine from the above periodic table. Do this by looking up sodium (Na) on the periodic table. Molar Mass of Ammonia NH 3 Step 1: The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Astatine is radioactive and rapidly decays to other, more stable elements. All of them are relatively common on Earth except for astatine. Now in MgCl2, there is 1 Magnesium atom and 2 Chlorine atoms. As you can see in the periodic table shown in the figure below, the halogens include the elements fluorine (F), chlorine (Cl), bromine (Cl), iodine (I), and astatine (At). The lightest chemical element is Hydrogen and the heaviest is Hassium. You can see the molar mass value of all the atoms from this periodic table. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium, Oxygen and Hydrogen from the above periodic table. 
Now in NaOH, there is 1 Sodium atom, 1 Oxygen atom and 1 Hydrogen atom. Now here we have to find the molar mass of NH3 (Ammonia). You can see the molar mass value of all the atoms from this periodic table. Because the molar mass of any molecule (or compound) can be calculated by simply adding the molar masses of individual atoms. This list contains the 118 elements of chemistry. If you have a periodic table with you, then you can easily calculate the molar mass of NH3 (Ammonia). There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. Visualize trends, 3D orbitals, isotopes, and mix compounds. Interactive periodic table showing names, electrons, and oxidation states. Weve talked about it in other videos, you could view this 12. Calculator - the best online scientific calculator. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. So if we first look at carbon, carbon, we see from this periodic table of elements, has a molar mass of 12.01 grams per mole.Now here we have to find the molar mass of O2 (Oxygen). The atomic mass (atomic weight) values cited on a periodic table are weighted averages of naturally occurring isotopes. 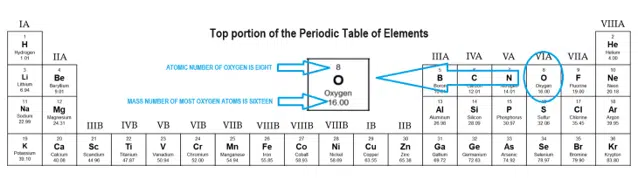
Plant Inspection & Process Optimalisation If you have a periodic table with you, then you can easily calculate the molar mass of O2 (Oxygen). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
